
Robert K. S. Wong, PhD
Distinguished Professor and Chair
Physiology and Pharmacology
Neurology
Our experiments are designed to examine the functional organization of the hippocampus. The properties of synaptic interactions mediated by the neurotransmitters GABA and glutamate are studied in some detail in order to understand how these interactions affect the overall network output.
Studies on the cellular and molecular aspects of neurons have contributed importantly to our understanding of the brain. A full description of the structure and function of the brain requires an examination of how interactions between individual elements bring about the particular behavior of the population of neurons. For example, our studies on the activities in the glutamatergic synapses between the pyramidal cells in the CA3 region give rise to a hypothesis regarding the reverberation of neuronal activities in a network. These reverberating activities can form the substrate for memory or give rise to seizure discharges in epilepsy. Fine tuning of the interaction between the glutamatergic synapses is provided by the inhibitory synaptic transmission mediated by GABA. Our studies show that the GABA receptors are regulated by intracellular calcium and phosphorylation. In addition, a novel excitatory synaptic action of GABA between inhibitory neurons has been uncovered in our experiments, providing insights on the signaling process within the inhibitory network in the hippocampus. Activity-dependent changes in the inhibitory and excitatory synapses are important issues that we are pursuing for a full understanding of the adaptive changes occurring in the brain that underlie all its fundamental functions.
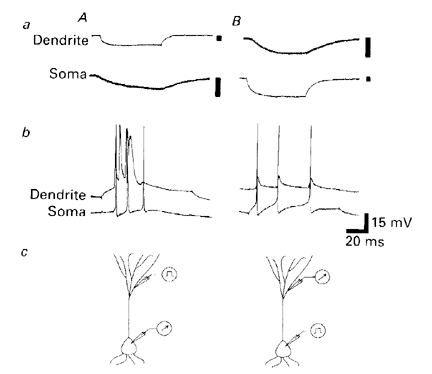
Figure 1. Coupling of dendritie and somatic intracellular recordings. In each of the four paired recordings, the dendritic response is shown above the somatic response. Aa, Hyperpolarizing current pulse injected through the dendritie recording electrode and the passive response recorded at the soma. Ba, hyperpolarization applied at soma is electrotonically conducted to the dendritic recording site. Ab, Depolarization of the apical dendrite elicits a local burst response consisting of fast and slow components. The response of the soma is a burst of fast action potentials. Bb, Depolarization of the soma produces repetitive firing. Attenuated spikes appear at the dendritic location. The drawings at the bottom (Ac and Bc) indicate which element was stimulated (apical dendrite in A; soma in B). Calibrations: vertical bars in top four traces (A and B, dendrite and soma) all 10 mV; time calibration same as bottom four traces: 20 ms. (From Wong and Stewart, 1992)
PersonnelAbraham Chuang, BSc, PhD, Research Assistant Professor
Steve Young, PhD, Research Instructor
Wangfa Zhao, BS, PhD, Research Assistant Professor
Reviewer for various scientific publications.
Membership on 3 NIH Study Sections
- Traub R.D. & Wong R.K.S. Cellular mechanism of neuronal synchronization in epilepsy. Science 216: 745-747 (1982).
- Miles R. & Wong R.K.S. Single neurones can initiate synchronized population discharge in the hippocampus. Nature 306: 371-373 (1983).
- Miles R. & Wong R.K.S. Latent synaptic pathways revealed after tetanic stimulation in the hippocampus. Nature 329: 724-726 (1987).
- Traub R.D., Miles R. & Wong R.K.S. Model of the origin of rhythmic population oscillations in the hippocampal slice. Science 243: 1319-1325 (1989).
- Michelson H.B. & Wong R.K.S. Excitatory synaptic responses mediated by GABAA receptors in the hippocampus. Science 253: 1420-1423 (1991).
- Zhong, J, Chuang S.C., Bianchi,R., Zhao W., Lee H., Fenton A., Wong, R.K.S. and Tiedge H. BC1 regulation of metabotropic glutamate receptor-mediated neuronal excitability. J. Neurosci. 29: 9977 - 9986 (2009).
- Osterweil EK, Chuang S-C, Chubykin AA, Sidorov M, Bianchi R, Wong RK, Bear MF. Lovastatin corrects excess protein synthesis and prevents epileptogenesis in a mouse model of fragile X syndrome. Neuron 77: 243-250 (2013).
- Young SR, Chuang S-C, Zhao W, Wong RK* and Bianchi R*. Persistent Receptor Activity Underlies Group I mGluR-Mediated Cellular Plasticity in CA3. * co-senior authors. J Neurosci. 33:2526-2540. (2013)
- APP Causes Hyperexcitability in Fragile X Mice. Westmark CJ, Chuang SC, Hays SA, Filon MJ, Ray BC, Westmark PR, Gibson JR, Huber KM, Wong RK. Front Mol Neurosci. 15;9:147. doi: 10.3389/fnmol.2016.00147 (2016)
- Kazim SF, Chuang SC, Zhao W, Wong RK, Bianchi R, Iqbal K. Early-Onset Network Hyperexcitability in Presymptomatic Alzheimer's Disease Transgenic Mice Is Suppressed by Passive Immunization with Anti-Human APP/Aβ Antibody and by mGluR5 Blockade. Front Aging Neuroscience 9:71. doi: 10.3389/fnagi.2017.00071 (2017).